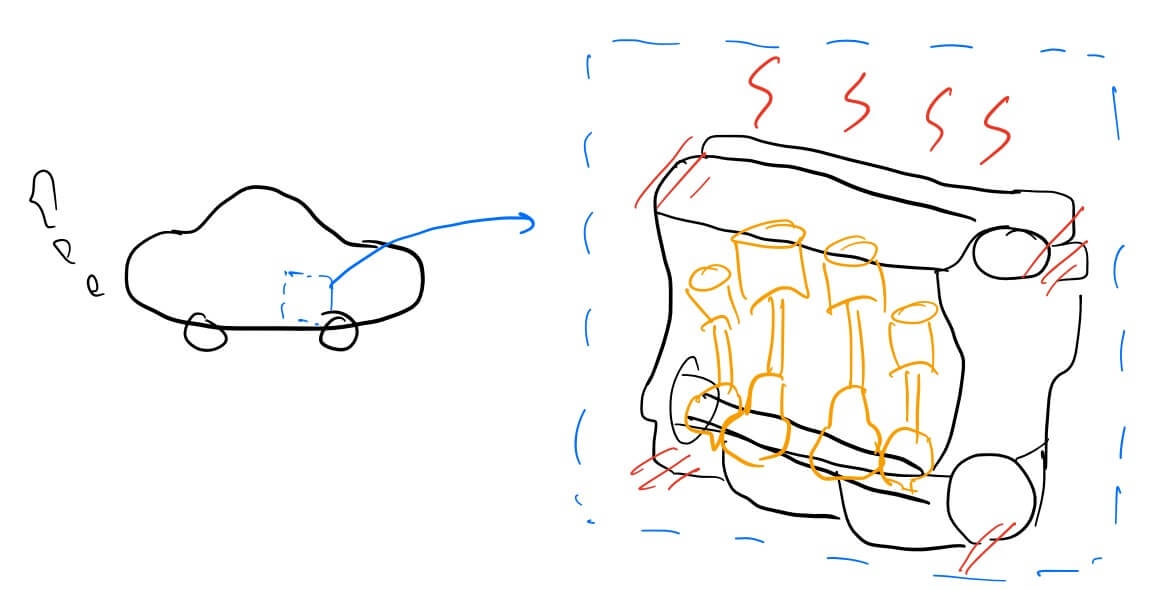
Heat transfer three points
- why the energy eventually return to heat → entropy, thermodynamics 2nd law, freedom, natual state
- how the external energy works → create order (lower the entropy) or raising the temperature
- why heat transfer defines product performance → waste, reduce performance, efficiency, deform permanently
All energy eventually converges into heat (The final destination of energy)
We create energy from various natural sources, such as fossil fuels (oil/coal) and electrical energy (including eco-friendly energy). This energy powers machines and devices, making them functional.

Engineers strive to maximize the efficiency of this energy. This is because wasted or “discarded” energy inevitably occurs during energy consumption—there is no such thing as 100% efficiency. Much of this energy transforms into heat, which becomes unusable natural energy, or is lost through noise and vibration. Ultimately, all energy disperses into nature as thermal energy. For instance, light eventually scatters as heat or hits our skin, minutely raising its temperature.

Why is heat generated? (Random Kinetic Energy and the Atomic Spring Model)
Heat = Random Kinetic Energy
Heat can be described as Random Kinetic Energy. To understand, one must look at the atomic level. Objects are made of countless atoms bonded together. Throwing an object means all these atoms move at the same velocity. However, if atoms move back and forth in the same direction, it becomes vibration; if they move randomly and collide continuously, it becomes heat.
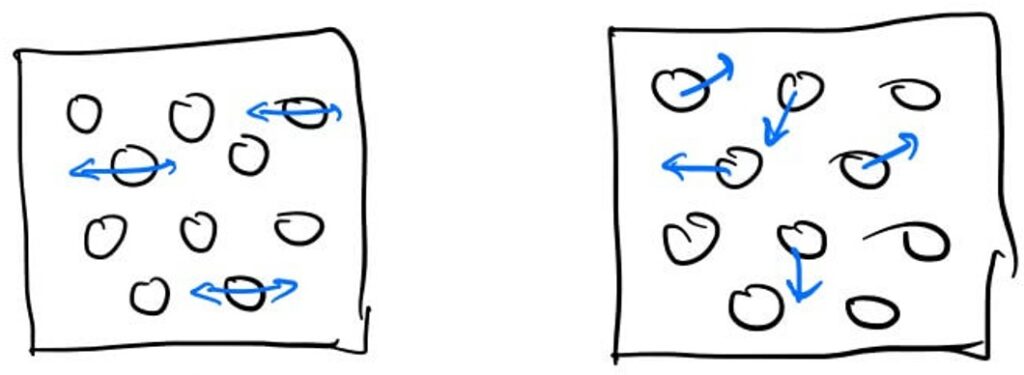
This is the origin of thermal energy. Natural phenomena tend toward this random and free form rather than order. Thus, energy that we have “ordered” for our use eventually regains its freedom and scatters as heat. When you hit an object with force, energy is transferred outward through sound or movement, but the remaining random vibrations within the object stay as thermal energy.

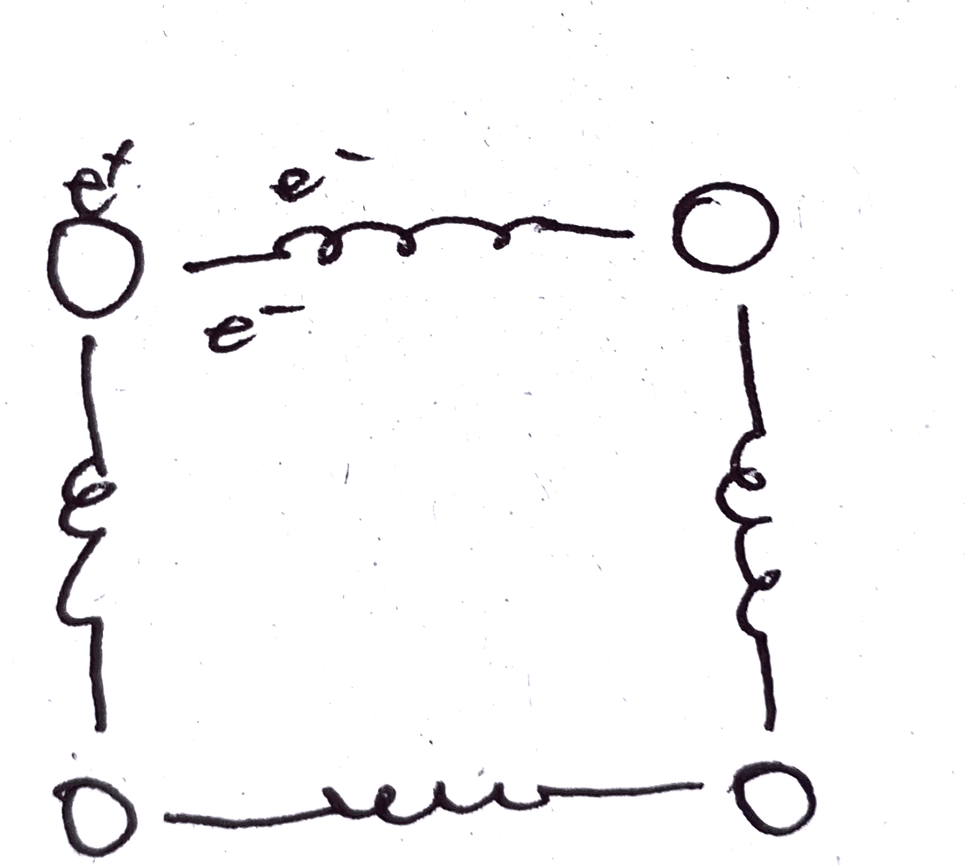
Material property, heat capacity (explain with spring and atom)
Atomic bonds are often compared to springs. If we view atoms as tiny spheres with mass and light spring, even a small force can create large random vibrations. When a significant force acts on a small-mass sphere connected by weak springs, its velocity changes in real-time.
In physics and chemistry, the relationship between bonding energy and distance is often represented by a graph.
- Point O: The most stable position where atoms stay with minimal energy.
- Energy E: The energy required to compress or stretch the bond.
- Potential h: Represents the “strength” of the spring.
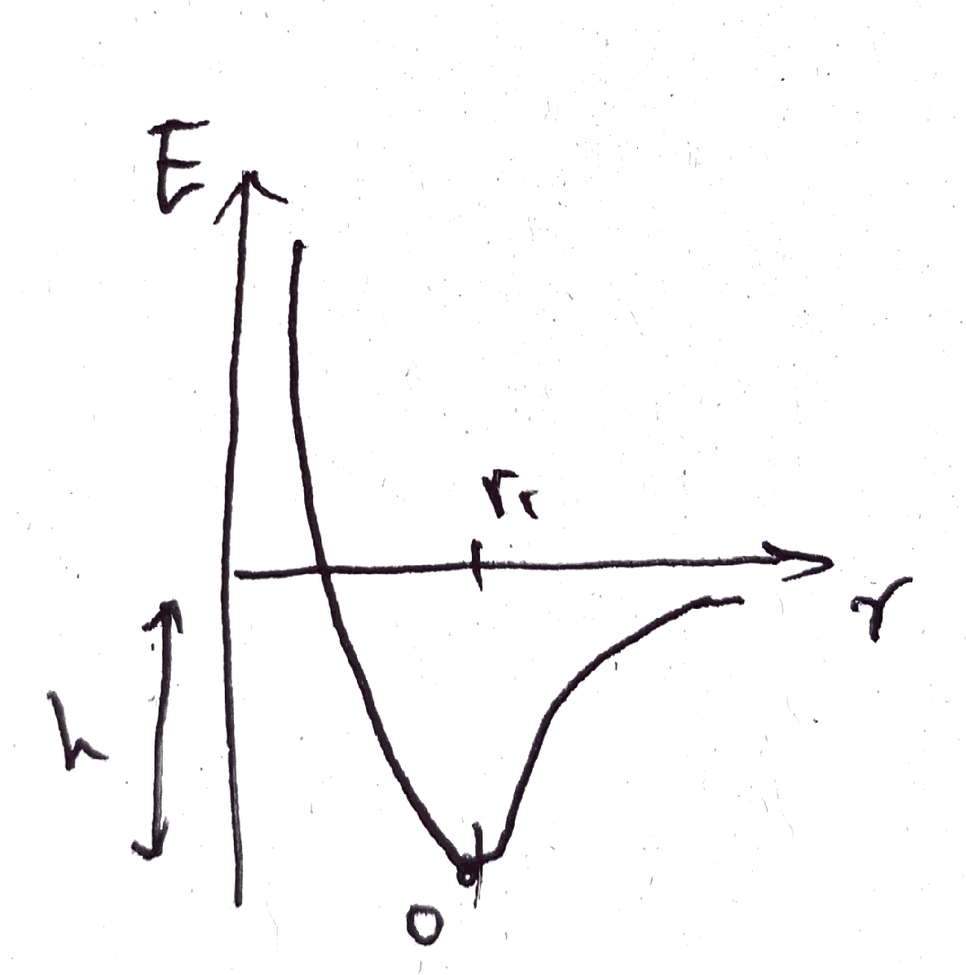
A high h indicates a strong spring. Strong springs don’t move much under small forces, but under large forces, they create many fine vibrations rather than swinging broadly. This relates to material properties like heat capacity and thermal conductivity. Materials with “stronger” atomic springs may generate more heat under large forces but have a high heat capacity, meaning their temperature rises slowly. Conversely, “weaker” springs allow atoms to gain random kinetic energy easily, leading to a lower heat capacity where temperature rises and falls quickly.

The Relationship Between Temperature and Freedom
Heat = Random Kinetic Energy
Since heat is random kinetic energy, it is difficult to eliminate all motion exists. Unless we reach absolute zero (a purely theoretical state), some degree of movement always persists. Even in a stationary object, molecular torsions cause atoms to hit neighbors, transferring energy in a continuous cycle.
Since heat is random kinetic energy, it is difficult to eliminate as long as atomic motion exists. Unless we reach absolute zero (a purely theoretical state), some degree of movement always persists. Even in a stationary object, molecular torsions cause atoms to hit neighbors, transferring energy in a continuous cycle.
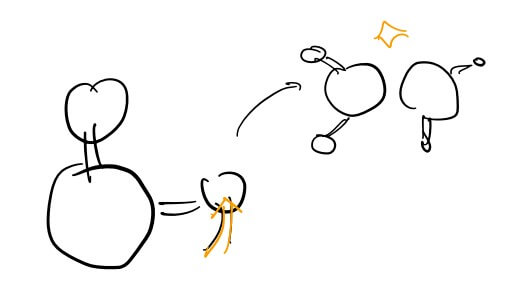
This also explains why temperature and pressure are proportional in gases. Increasing pressure reduces volume, forcing molecules to move faster and collide more frequently, which generates heat and raises the temperature
Temperature vs. Heat: The Level and the Energy
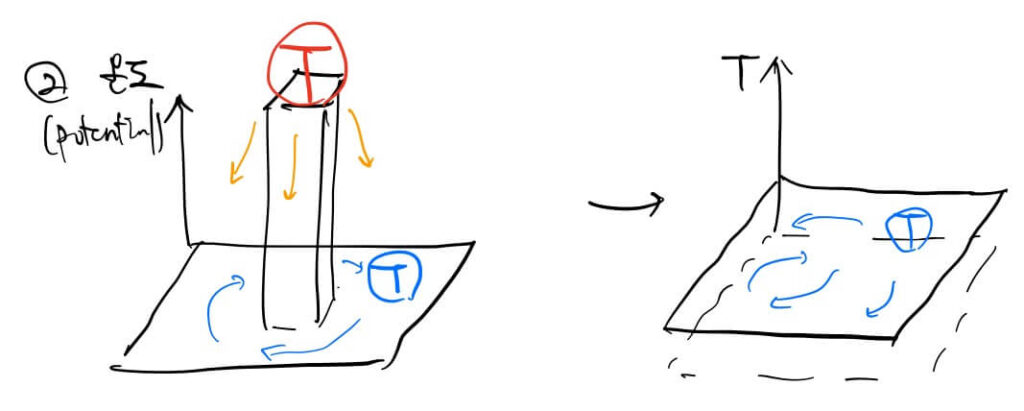
Simply put, Temperature is a “level” or “potential,” while Heat is the energy that can raise that level. Just as electricity flows from high voltage to low voltage, thermal energy flows from high temperature to low temperature.
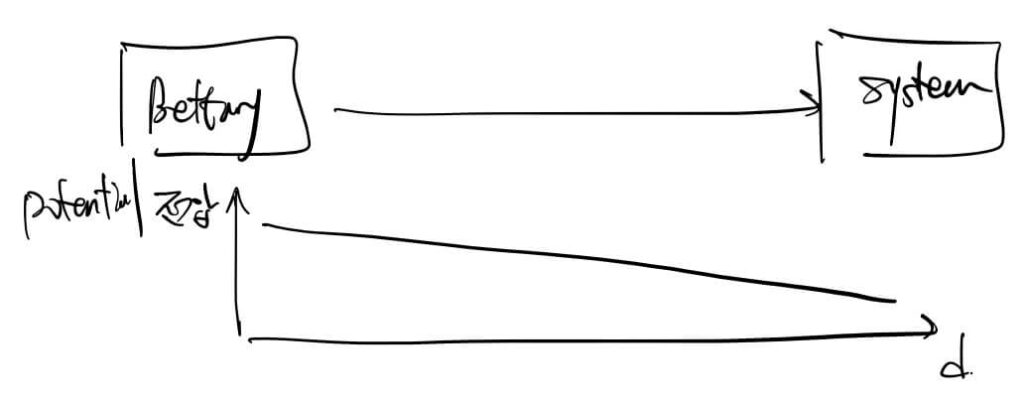
This relationship involves Degrees of Freedom (Entropy). The Second Law of Thermodynamics states that entropy always increases in an isolated system. Nature moves toward disorder and higher freedom. Energy transitions from a useful, refined state to a useless, scattered state (heat).
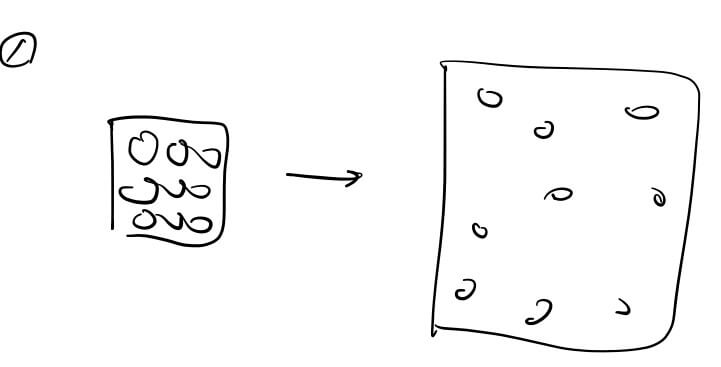
We can summarize the relationship as:
Internal Energy (Thermal Energy) = Entropy + Temperature
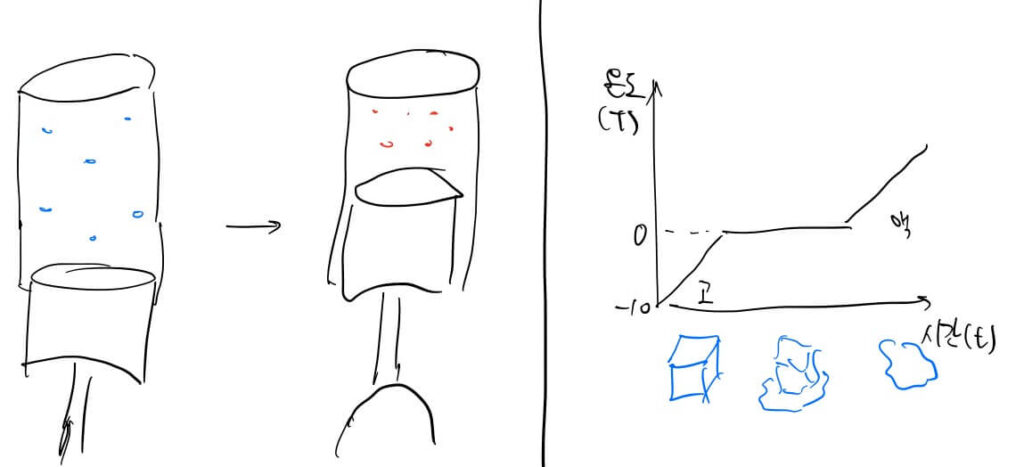
- When external energy is added: Energy can be used to create order (lower entropy) while raising temperature. For example, compressing air in an engine requires external energy and raises the temperature.
- Phase Change: When energy is used entirely to increase freedom (breaking bonds/expanding space), the temperature does not rise despite the energy input.
- When no external energy is added: A system will try to equalize temperature potentials to maximize freedom. A hot bowl of ramen in a cold room eventually disperses its heat until it matches the room’s temperature, increasing the overall entropy of the system.
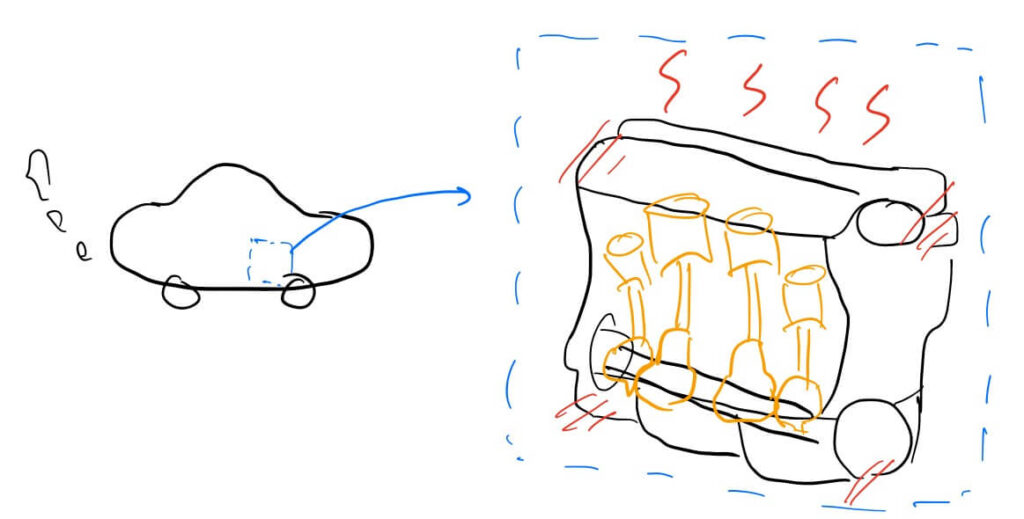
Why Heat Transfer Defines Product Performance
Electronic devices like laptops and smartphones heat up as they consume power. If this heat is not discharged, it causes performance “throttling” and can lead to permanent damage

The same applies to electric motors in EVs or industry. Excessive heat means the input energy is being wasted instead of doing work, reducing efficiency. Furthermore, high temperatures can cause magnets to lose their magnetism or coatings to deform permanently.
From an engineer’s perspective:
- First Priority: Design a system with high energy conversion rates to minimize heat generation.
- Second Priority: Efficient method that distribute the heat to protect the performance and lifespan of the product is important
three points
- why the energy eventually return to heat → entropy, thermodynamics 2nd law, freedom, natual state
- how the external energy works → create order (lower the entropy) or raising the temperature
- why heat transfer defines product performance → waste, reduce performance, efficiency, deform permanently
- wiki
EV Powertrain Engineer specializing in Motors and Reducers (gear box). Sharing hands-on industry experience and engineering insights—spanning from R&D design to Thermal, Fluid, and Structural analysis.
↓
Key Expertise
M.S. in Heat Transfer lab [Mar 2020 ~]
EV Motor Stator Assembly Design & Motor Performance Test and Analysis [Apr 2022 ~]
Gearbox Lubrication CFD Analysis & Lubrication/Performance Testing [Jan 2024 ~]
Powertrain Performance Testing & Data Analytics [Feb 2026 ~]